Organisation at a glance
Our Vision
Saving and improving Australian lives through a world-class blood supply.
Our Role
The National Blood Authority (NBA) is a statutory agency within the Australian Government Health and Ageing portfolio that manages and coordinates arrangements for the supply of blood and blood products and services on behalf of the Australian Government and state and territory governments.
The key role of the NBA is to:
- provide an adequate, safe, secure and affordable supply of blood products, blood related products and blood related services, and
- promote safe, high quality management and use of blood products, blood related products and blood related services in Australia.
The NBA:
- works with jurisdictions to determine the clinical requirements for blood and blood products and develop an annual supply plan and budget
- negotiates and manages national contracts with suppliers of blood and blood products to obtain the products needed
- assesses blood supply risk and engages in contingency planning for risks arising in the sector
- supports the work of the jurisdictions to improve the way blood products are used - including developing and facilitating strategies and programs that will improve the safety, quality and effectiveness of blood usage, particularly in the areas of national standards, guidelines and data capture and analysis
- provides expert advice to support government policy development, including identification of emerging risks, developments, trends and new opportunities
- manages the evaluation of proposals for blood sector improvements, including proposals for new products, technologies and system changes
- provides secretariat support to the Jurisdictional Blood Committee (JBC).
Authority
The NBA was established by the National Blood Authority Act 2003 following the signing of the National Blood Agreement by all state and territory health ministers in November 2002. As a material statutory agency, the NBA has a range of corporate and compliance responsibilities under the National Blood Authority Act 2003, the Financial Management and Accountability Act 1997, and the Public Service Act 1999, along with a responsibility to meet ministerial, parliamentary and financial reporting requirements.
Responsible Ministers and Portfolio
The NBA exists within the portfolio responsibilities of the Minister for Health. The NBA General Manager is a statutory officer who reports to the standing committee on Health (SCoH) and the Commonwealth Minister for Health.
Our Outcome
Access to a secure supply of safe and affordable blood products, including through national supply arrangements and coordination of best practice standards within agreed funding policies under the national blood arrangements.
Funding
Under the National Blood Agreement between the Australian Government and the states and territories, 63 per cent of NBA funding is provided by the Australian Government and the remaining 37 per cent is provided by the state and territory governments. The funding covers both the national blood supply and the operations of the NBA.
Since the establishment of the NBA, governments have funded a total of $7,623.3 million on blood and blood products. In 2012-13, the total amount provided to cover expected demand for blood and blood products was $1,049.3 million.
TABLE 1.1 Government funding for the supply of blood and blood products, 2003-04 to 2012-13
| Year | Amount ($M) | Growth (%) |
|---|---|---|
| 2003-04 | 460.5 | |
| 2004-05 | 536.8 | 16.6 |
| 2005-06 | 577.4 | 7.6 |
| 2006-07 | 639.4 | 10.7 |
| 2007-08 | 719.5 | 12.5 |
| 2008-09 | 806.8 | 12.1 |
| 2009-10 | 878.8 | 8.9 |
| 2010-11 | 939.2 | 6.9 |
| 2011-12 | 1,015.6 | 8.1 |
| 2012-13 | 1,049.3 | 3.3 |
| Total | 7,623.3 | 9.6 (average) |
TABLE 1.2 Government funding for the operation of the NBA, 2003-04 to 2012-13
| Year | Amount ($M) | Growth (%) |
|---|---|---|
| 2003-04 | 7.4 | |
| 2004-05 | 8.4 | 13.5 |
| 2005-06 | 10.4 | 23.8 |
| 2006-07 | 10.1 | -2.9 |
| 2007-08 | 9.6 | -5.0 |
| 2008-09 | 9.2 | -4.2 |
| 2009-10 | 8.9 | -3.3 |
| 2010-11 | 9.5 | 6.7 |
| 2011-12 | 8.5 | -10.5 |
| 2012-13 | 10.3 | 21.2 |
| Total | 92.3 | 4.4 (average) |
Our Staff
As at 30 June 2013, the NBA had 45 ongoing and 11 non-ongoing staff.
Location
The NBA staff are located in Canberra at Level 2, 243 Northbourne Avenue, Lyneham ACT.
Key Events in the NBA’s History
| 2003 | Established by the National Blood Authority Act 2003 following the signing of the National Blood Agreement by all state and territory health ministers in November 2002 |
| 2006 | NBA executed a Deed of Agreement with the Australian Red Cross Society for the provision of fresh blood products |
| NBA won the Prime Minister’s Silver Award for Excellence in Public Sector Management for procurement of recombinant (manufactured) products | |
| 2007 | First edition of Criteria for the clinical use of intravenous immunoglobulin (IVIg) in Australia was published |
| 2008 | Launch of the National Blood Supply Contingency Plan (NBSCP) to provide clear governance for managing blood shortages |
| Launch of the redeveloped Australian Bleeding Disorders Registry (ABDR) to better support planning and clinical management of people with bleeding disorders | |
| 2009 | Establishment of the Australian National Haemovigilance program to report on serious transfusion related adverse events |
| NBA was awarded with the Australian Government Comcover Award for Excellence in Risk Management for the NBSCP | |
| 2010 | New CSL Australian Fractionation Agreement came to effect |
| NBA won a United Nations Public Service Award in the Advancing Knowledge Management in Government category | |
| 2011 | National rollout of BloodNet, an online web based blood ordering system |
| Release of the first module (Critical Bleeding/Massive Transfusion) of the Patient Blood Management (PBM) Guidelines | |
| 2012 | Release of Module 2 Perioperative and Module 3 Medical of the PBM Guidelines |
| Second edition of IVIg Criteria in Australia was published | |
| 2013 | Release of Module 4 Critical Care of the PBM Guidelines |
Year at a Glance: Snapshot of the blood sector in 2012-13





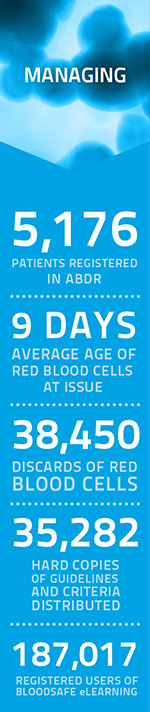


Key Achievements
PBM Guidelines: Medical and critical care modules released
2nd edition of IVIg criteria released
Subcutaneous immunoglobulin approved for national supply list
Cost indicator introduced on fresh blood product labels
Bloodnet: 342 sites, 95% national coverage
Managing blood and blood product inventory guidelines
2nd output based funding model and handover plan agreed with blood service
