In this Autumn 2019 issue:
- System Activity Update
- BloodSTAR 3.1 Release
- Changes to BloodPortal Password Expiry Requirements
- User Tips
- Changing a Patient’s Treatment Arrangements
- Removing and/or Editing an Individual Dispense Request
- Submitting Feedback in BloodSTAR
- Ig Governance Update
- Implementation of BloodSTAR in NSW Increases Blood Sector Systems Daily Use
System Activity Update
As of 30th April there were:
- 13,648 patients with active authorisations, and
- 13,764 registered users accessing BloodSTAR as Authorisers, Medical Officers, Nurses, Admin Support or Facility Administrators.
In April there were 938 initial authorisation requests and 16,950 dispense episodes of IVIg/SCIg in BloodSTAR nationally.
BloodSTAR 3.1 Release
BloodSTAR 3.1 was successfully released into production on 31 March 2019. This update included new functions and enhancements that improve the usability and functionality of the systems including:
- A warning message to Medical Officers if unused patient doses may be lost in a continuing treatment request.
- The ability to notify and request Medical Officers to change from a domestic Ig product to an imported product for specified patients and authorisations.
- The ability to delete a draft continuing authorisation request, and
- A requirement to add a contact name and phone number when creating/updating a planning sheet or a dispense request.
Changes to BloodPortal Password Expiry Requirements
In line with Australian Government Information Security Manual (ISM) requirements, the NBA will implement a number of changes to password management for the BloodPortal.
These changes include:
- Implementing a mandatory minimum password length of 10 characters with at least three of the following character sets
- lowercase alphabetic characters (a-z)
- uppercase alphabetic characters (A-Z)
- numeric characters (0-9)
- special characters
- Requirement for passwords to be changed every 90 days, and
- Prevent passwords from being used within eight password changes.
The NBA is aiming to migrate all users to the new requirement by 1 July 2019 (except NSW BloodSTAR users). The NBA is currently working with NSW to evaluate logon options for NSW BloodSTAR users.
We thank all our users for understanding as we implement these new requirements. For further information and for any feedback on this matter please contact us on 13 000 BLOOD (13 000 25663) or support@blood.gov.au.
User Tips
Changing a patient’s treatment arrangements
When a patient changes facility, you can update their treatment arrangements on their authorisation record. This functionality is available for all BloodSTAR users with Medical Officer, Nurse and Administration Support access. To update a patient’s treatment arrangements:
- Locate the patient from your home page or via the ‘Search’ page and click on their authorisation
- number.
- Under the authorisation details tab an ‘Edit’ button will display – click on the button and scroll to the bottom of the page to find the patient’s listed treatment arrangements.
- Update the required fields and click ‘Save’ to finalise.
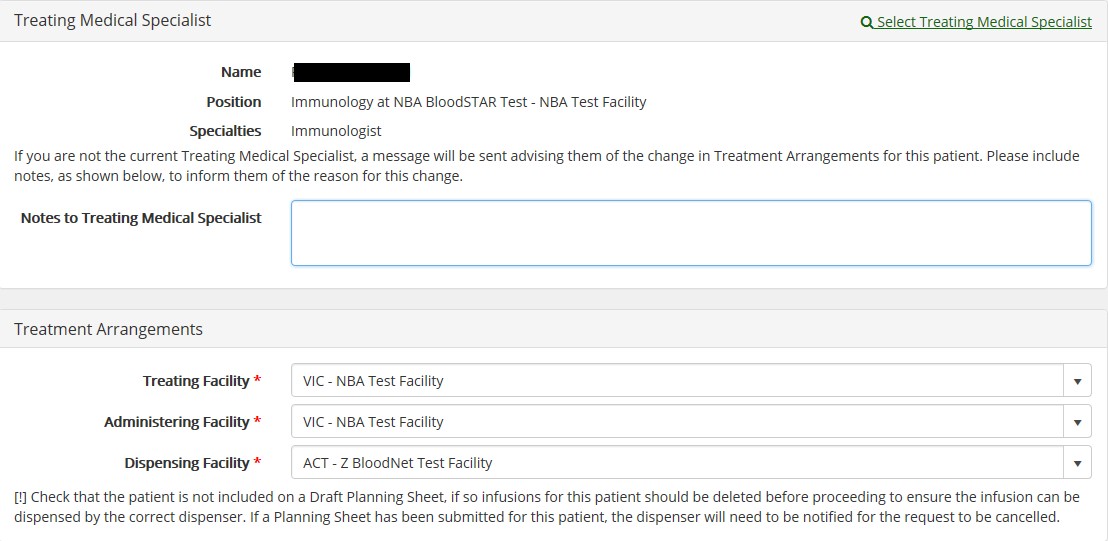
Further information on updating a patient’s treatment arrangements is available in this BloodSTAR tip sheet.
Removing and/or editing an individual dispense request
After an individual dose request has been submitted but not yet dispensed, Medical Officers, Nurses and Administration Support have the ability to remove or edit the request. This may be required if an incorrect expected infusion date is entered, or if a patient’s scheduled treatment date is changed or cancelled.
Editing a dispense request
- Click on the treatment tab and select ‘Planning Sheets’ from the dropdown menu.
- Locate the individuals request and click on the ‘View’ hyperlink beside it.
- From the request select the ‘Edit’ hyperlink to display the edit dispense request page.
- To change the expected infusion date or dose quantity, click on the ‘Edit’ link under the dose details and amend and save.
The system will display a message to advise that you must contact your dispenser to inform them of the change to ensure the correct product and dose is dispensed.
Removing a dispense request
- Click on the treatment tab and select ‘Planning Sheets’ from the dropdown menu.
- Locate the individual’s request and click on the ‘View’ hyperlink beside it.
- Next to the request details, select the ‘Remove’ hyperlink, which will display a message to confirm the removal of the request.
- After confirming the removal, the system will display a message to advise that you must contact your dispenser to ensure they don’t supply the removed dose.
Submitting feedback in BloodSTAR
You can now provide feedback on BloodSTAR or the Criteria by selecting ‘Feedback’ in the menu bar at the top of any page.
![]()
We welcome feedback to assist us with our commitment to improving our service. If you’ve had a positive experience with BloodSTAR or you have a suggestion on how we can improve this system please let us know on 13000 BLOOD (13 000 25663) or support@blood.gov.au.
Ig Governance Update

The Criteria for Immunoglobulin Use in Australia (the Criteria) have changed to Version 3. These changes have been in effect in BloodSTAR since 22 October 2018 through the release of BloodSTAR version 3.0.
Version 3 of the Criteria aim to more clearly articulate and standardise diagnostic, qualifying and review requirements. More comprehensive justification and evidence from clinicians will be required to confirm patient eligibility and so will strengthen the capacity to direct Immunoglobulin (Ig) product to those who most require it. National funding of Ig products is no longer supported for a small number of low-use conditions as better alternative treatments are available.
As part of the Version 3 Criteria, some existing authorisations will require the Medical Officer to provide some additional historical information about the patient’s diagnosis and initial response to Ig therapy to ensure the patient is still eligible for Ig therapy and is authorised under the correct indication. Since 22 October 2018, 4,504 patients have been successfully transitioned from Version 2 to Version 3 of the Criteria across Australia.
It’s important to note that the Criteria are not intended to be clinical practice guidelines. Rather, the Criteria identify the conditions and circumstances for which the use of Ig products is considered to be clinically appropriate and for which Ig products are able to be accessed under the National Policy. Prescribers should follow local requirements for prescribing Ig products in accordance with relevant legislation.
For more information regarding Version 3 of the Criteria, including responses to some common questions, please visit the Version 3 Criteria Newsletter Update.
For the latest information on the Ig Governance program please visit https://www.blood.gov.au/Ig-program- updates.
Implementation of BloodSTAR in NSW Increases Blood Sector Systems Daily Use
With the October 2018 implementation of BloodSTAR in NSW, the NBA’s Blood Sector Systems now support over 15,000 users nationally.
Here is a table of the 2018 monthly user statistics.
| Month | ABDR | MyABDR | BloodSTAR | BloodNet |
| Jan | 90 | 266 | 1810 | 2667 |
| Feb | 88 | 283 | 1816 | 2646 |
| Mar | 91 | 294 | 1824 | 2665 |
| Apr | 80 | 303 | 1808 | 2647 |
| May | 99 | 327 | 1929 | 2687 |
| Jun | 85 | 356 | 1850 | 2665 |
| Jul | 90 | 394 | 1862 | 2739 |
| Aug | 100 | 388 | 1992 | 2735 |
| Sep | 84 | 371 | 1847 | 2655 |
| Implementation of BloodSTAR in NSW | ||||
| Oct | 92 | 380 | 2749 | 2821 |
| Nov | 97 | 385 | 2829 | 2830 |
| Dec | 95 | 391 | 2632 | 2772 |
For Further Information
Further information on BloodSTAR is available online at www.blood.gov.au/bloodstar.
Previous Editions
To read or download previous editions of BloodSTAR News, click on the relevant link below:

