Labelling of blood and blood products
Barcode symbology on blood and blood product labels is changing. This affects products funded under the national blood arrangements.
Australia is moving to the globally recognised label standards. Adopting these barcodes:
- improves traceability in the blood and blood product supply chain, which enhances safety and supply security
- improves inventory management and financial sustainability
- ensures that blood products are given a unique and unmistakable identification, which increases efficiencies
- provides internationally standardised product codes and definitions for blood components, which facilitates global compliance and benchmarking
- stops duplication of donation identification numbers (DIN) within a 100-year period worldwide.
These barcodes sit alongside human readable information on blood product labels.
Watch the Barcoding Reform Open Forum 2015(Opens in a new tab/window) video for more information about the move to global standards.
Since 2014, all blood components supplied by Australian Red Cross Lifeblood(Opens in a new tab/window) (Lifeblood) also include a manufacturing cost indicator on the blood bag label.
Barcode specifications
Australia has adopted the following globally recognised standards for blood and blood product label barcodes:
- ISBT 128 2D DataMatrix(Opens in a new tab/window) – for all fresh blood products (red blood cells, platelets, clinical fresh frozen plasma, cryoprecipitate, cryodepleted plasma and serum eye drops)
- GS1 2D DataMatrix(Opens in a new tab/window) – for all plasma-derived, recombinant and diagnostic products.
Find out more about GS1 global label barcode standards in the Barcoding plasma derivatives implementation guide(Opens in a new tab/window).
The Barcode specifications for blood and blood products funded under the National Blood Arrangements sets out the specifications we require for blood product labels.
Lifeblood's Australian guidelines for the labelling of blood components using ISBT 128(Opens in a new tab/window) give specific information about the transition to the ISBT 128 label standard.
The GS1 General Specifications Standard(Opens in a new tab/window) defines how to use identification keys, data attributes and barcodes in business applications of the GS1 system standards.
Status of commercial blood product labelling
By the end of 2024, CSL Behring blood product labels will only include the GS1 2D DataMatrix barcode and human-readable information. Find out more about the transition of Australia's domestic plasma products.
All other commercial suppliers are using either GS1 linear or GS1 2D DataMatrix barcodes or both, plus human-readable information.
We are still investigating the need for serialisation. This will be included in a project to develop an electronic blood-tracking policy.
Suppliers of manufactured batch products
It is mandatory for blood and blood product manufacturers to transition to the new barcode symbology.
For more information about GS1 2D DataMatrix transition, read the barcoding FAQs for suppliers of manufactured batch products and the FAQ below:
We publish samples of barcodes created by suppliers as they become available.
Status of fresh blood product labelling
On 18 November 2018, Lifeblood introduced a transition label, including the Codabar barcode and ISBT 128 linear barcode.
All barcodes for fresh blood products must transition completely to the ISBT 128 2D DataMatrix. The 2D ISBT 128 DataMatrix was implemented from 30 November 2025.
The ISBT 128 linear barcodes and eye-readable information appearing in the upper portion of the current label have not changed. The contact phone number has been updated.
The transition label with the Codabar barcodes will be removed on 31 March 2029. After that time, labels will no longer include the Codabar barcode.
What Australian Health Providers need to know
You may need to update your laboratory information systems and infrastructure to process the new labels. This includes barcode scanning equipment. In November 2023, we provided all Australian Health Providers with 2 years notice to allow time for this transition.
On 27 November 2023, we sent a letter to all Australian Health Providers who manage blood and blood products. This noted a transition period for changes to Lifeblood's blood component label. This applies to all fresh blood products funded under the national blood arrangements.
Lifeblood implemented a transition label in November 2018. It included a new ISBT 128 linear barcode, the existing Codabar barcode and human-readable information in distinct sections. This transition label helped facilities manage inventory without disrupting blood supply.
From 30 November 2025, the ISBT 128 2D DataMatrix was added to existing fresh blood product competent label. The addition of the 2D DataMatrix will allow Australian Health Providers who have updated their laboratory information systems to begin using the best practice barcodes.
On 31 March 2029, the Codabar barcode will be removed from fresh blood product component labels. All updated labels will include:
- the new ISBT 128 2D DataMatrix
- the ISBT 128 linear barcode
- human-readable information.
The ISBT 128 linear and 2D DataMatrix barcodes represent best practice for blood product labelling. We can encode the same information contained in multiple linear codes into the 2D DataMatrix as a single ISBT 128 barcode. This technology will prepare us for the future and allow more rapid scanning of blood components at the point of issue and receipt.
We have now given Australian Health Providers over 6 years to update their laboratory information systems and infrastructure to process the new labels.
The Codabar barcode will not be available after 31 March 2029. If your facility is still using the Codabar barcode, you need to update your system and infrastructure, including barcode scanners, to read the ISBT 128 linear or 2D DataMatrix barcodes.
Lifeblood have provided a sample updated ISBT 128 fresh blood component labels with the 2D DataMatrix barcode. You can use this to help upgrade your systems and barcode scanners. The sample barcodes are available below for testing.
The new physical label will be 100 x 100 mm in size after the Codabar barcode is removed. The ISBT 128 linear barcode and human-readable information will remain in the upper portion of the label. A 2D DataMatrix will be added. Its placement on the label is being finalised.
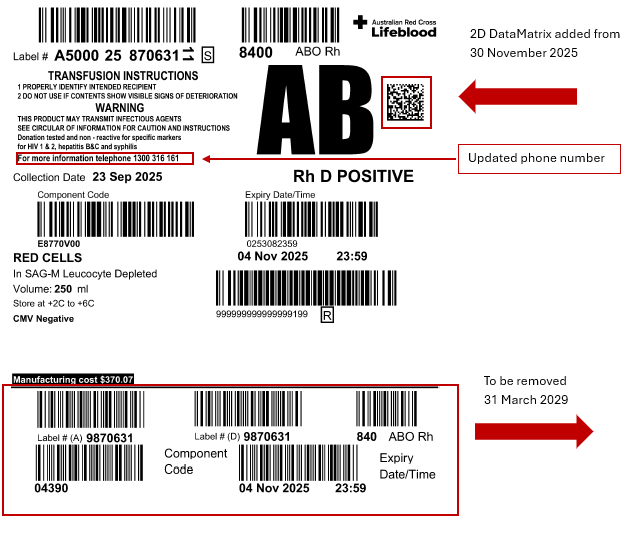
Example of an ISBT 128-compliant blood component transition label that includes ISBT128 linear and 2D, eye-readable, updated contact number and the transition barcode section.
You can use the ISBT 128 linear barcode without using the 2D DataMatrix barcode. If your facility already uses the ISBT 128 linear barcode, you can continue using it and choose not to update your system.
Note the implementation date relates to all fresh blood components coming off the manufacturing line and issued to Australian Health Providers. There will be a crossover period where an inventory of blood products may have both current and new labels.
Sample fresh blood component labels
Sample ISBT 128 fresh blood component labels with the 2D DataMatrix are now available for testing.
Red Blood Cells
Irradiated Pooled Platelets
Fresh Frozen Plasma
Help with transition
If you need help implementing the ISBT 128 2D DataMatrix standard, contact your local Transfusion Medicine Scientist or support@blood.gov.au.
Detailed information about ISBT 128 is available in the Australian guidelines for the labelling of blood components using ISBT 128(Opens in a new tab/window).
Technical information about ISBT 128 is available on the ICCBBA website(Opens in a new tab/window).
Manufacturing costs on labels
Fresh blood components supplied by Lifeblood include a manufacturing cost on the blood bag label. We calculate the indicative cost for each blood component type on an annual basis.
We introduced this initiative in 2014. It aims to help Australian Health Providers understand the costs of providing blood and blood products in Australia.
Blood donors in Australia don't receive payments for their donations. However, it still costs money to produce blood and blood products. This includes the costs of:
- collecting
- testing
- processing
- distributing.
These processes help us ensure the safety and quality of blood and blood products.
The Australian, state and territory governments cover the costs of producing blood and blood products in Australia. This is why blood and blood products are provided at no cost to patients when they need it.
Patients are never charged for the blood they receive, whether they are treated in public or private hospitals. The manufacturing cost on the label is not a cost to the patient. Instead, it serves as a reminder that blood is a precious resource generously provided by donors. Careful use and management of blood can avoid unnecessary wastage.
A certain amount of wastage is inevitable and appropriate. Some blood and blood products have short shelf lives. These must meet strict storage specifications to ensure product quality. This particularly affects fresh blood products. Some discards are necessary to make sure products are available when and where they are clinically needed.

Example of a red blood cell component label from 2020 that includes a cost indicator.
Get in touch
We update this page as required.
For more information about blood product labelling, please contact us.
Phone: 1300 025 663 (13 000 BLOOD)
Last updated: 27 Mar 2024
